 Once CO exposure is discontinued, dissociation of COHb occurs and CO is excreted through the lungs. Although the teratogenicity of CO is controversial, the risk of foetal injury is increased. The excessive left shift of foetal COHb curve makes tissue hypoxia more severe by releasing less oxygen to the foetal tissues. The final COHb levels in the foetus significantly exceeds the level in the mother. Carbon monoxide exposure has an especially deleterious effect on pregnant women, because of the greater sensitivity of the foetus to the harmful effects of the gas. The net result is cognitive defects, particularly in memory and learning with movement disorders that may not appear for days following the initial poisoning. With reperfusion of the brain, leukocyte adhesion and the subsequent release of destructive enzymes and excitatory amino acids all amplify the initial oxidative injury. The end result is lipid peroxidation of the brain, which starts during recovery from carbon monoxide poisoning. The release of nitric oxide from platelets and endothelial cells, which forms the free radical peroxynitrite, can further inactivate mitochondrial enzymes and damage the vascular endothelium of the brain. When carbon monoxide binds to cytochrome oxidase, it causes mitochondrial dysfunction resulting into oxidative stress related damage. 
Research suggests that the intracellular uptake of carbon monoxide is an important mechanism for neurologic damage. But the toxic effects of CO cannot be explained by these processes alone, as COHb levels do not correlate well with symptoms, outcome or the phenomenon of delayed neurologic sequelae. 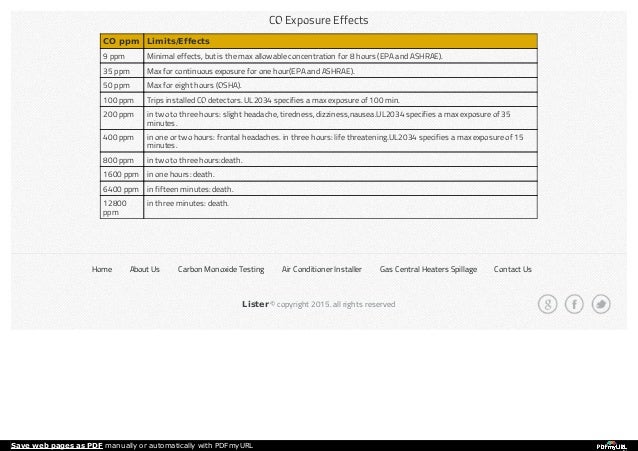
The resulting decrease in arterial oxygen content and shift of the oxyhemoglobin dissociation curve to the left explains the acute hypoxic symptoms (primarily neurologic and cardiac) seen in patients with acute poisoning. Carbon monoxide quickly binds with hemoglobin with an affinity greater than that of oxygen to form COHb. 
The amount of CO absorbed by the body depends on minute ventilation, duration of exposure and concentration of CO in the environment. In such cases, peak COHb levels may be delayed and prolonged because of on going production of CO from liver. People who work with methylene chloride as paint stripper, can be poisoned because the fumes are readily absorbed and converted to CO in the liver. It is a major contributor in the thousands of smoke inhalation deaths that occur each year. Ĭarbon monoxide can occur in the presence of other toxins, complicating management. In army setting poisoning is usually seen in high-altitude areas where unwary soldiers often sleep in closed tents with burning bhukharis (charcoal/kerosene) kept inside. 
Burning of charcoal, wood, kerosene, or natural gas for heating and cooking also produces carbon monoxide. According to a ten year review of carbon monoxide related deaths more than half of unintentional deaths were caused by motor vehicle exhaust. A smoker is exposed to 400 to 500 ppm of CO while actively smoking. The human body produces carbon monoxide as a byproduct of hemoglobin degradation, resulting in baseline carboxyhemoglobin (COHb) saturation of 1-3% in non-smokers, which increases to 10-15% amongst heavy smokers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
